PWA Duhl Advanced high
strength single crystal superalloy compositions (PWA 1484) 1988 US4719080A
Publication
Number: US4719080A
Publication
Date: 1988-01-12
Priority
Number: US1985743138A
Application
Date: 1985-06-10
Title:
Advanced high strength single crystal
superalloy compositions
Inventor
- w/address: Duhl David
N.,Newington,CT,US | Cetel Alan D.,West Hartford,CT,US
Assignee/Applicant:
United Technologies
Corporation,Hartford,CT,US
Front Page Drawing:
Abstract:
A superalloy composition and single crystal articles of the
composition are described. The broad range is 3-12% Cr, 0-3% Mo, 3-10% W, 0-5%
Re, 6-12% Ta, 4-7% Al, 0-15% Co, 0-0.045% C, 0-0.02% B, 0-0.1% Zr, 0-0.8% Hf,
0-2% Nb, 0-1% V, 0-0.7% Ti, 0-10% (Ru+Rh+Pd+Os+Ir+Pt), balance essentially Ni.
An equation is presented to select the most useful specific compositions from
within this range. An exemplary preferred composition is 5.0% Cr, 10.0% Co,
2.0% Mo, 6.0% W, 3.1% Re, 5.6% Al, 9.0% Ta, 0.1% Hf, balance essentially Ni.
Description
BEST MODE FOR CARRYING OUT THE INVENTION
Table I lists several
composition ranges of varying scope for the composition of the single crystals
of the present invention. All percent figures in this application are weight
percent figures unless otherwise indicated.
Nickel base
superalloys such as the invention composition are developed with certain
requirements in mind. Among these are oxidation resistance, corrosion
resistance and mechanical properties.
Resistance to
oxidation and corrosion is largely determined by the nature of the oxide layer
which forms on the alloy. In the invention composition the chromium and
aluminum act together to insure the formation of a protective oxide layer. The
layer contains substantial amounts of alumina but the alumina may be mixed with
other oxides depending upon the test conditions. If excessive amounts of
chromium are present, undesirable phases form unless other strengthening
elements are reduced. In addition to providing oxidation and corrosion
resistance the aluminum is the primary gamma prime former. The gamma prime
phase, Ni. sub.3 Al, is the phase which provides most of the strength of nickel
base superalloys. In addition to aluminum as a gamma prime former the present
alloy contains tantalum which is also a strong gamma prime former. These
elements are present in a sufficient amount to form from about 50 to about 75%
by volume of a gamma prime strengthening phase. When present, platinum,
palladium, ruthenium and osmium are also effective in increasing the oxidation
and corrosion resistance of the material. In many superalloys titanium and/or
vanadium are used as gamma prime formers. In the present alloy, however, it is
not desirable to employ titanium or vanadium as gamma prime formers since the
utilization of titanium or vanadium for this purpose will limit the amount of
aluminum which can be used as a gamma prime former and will thereby effectively
reduce the oxidation resistance of the material.
The refractory
elements, molybdenum, tungsten and rhenium, are present to act as solid
solution strengtheners and these elements primarily improve the properties of
the gamma matrix.
The invention
composition will be cast in single crystal form according to the teachings of
various prior art patents including U.S. Pat. Nos. 3,700,023; 3,763,926 and
4,190,094 which are incorporated herein by reference.
The compositions and
single crystal articles of the present invention will preferably be given a
heat treatment in accordance with the teachings of U.S. Pat. No. 4,116,723. For
alloy B49 (described in Table I) , the most promising alloy yet identified within
the scope of the present invention, the preferred heat treatment is a solution
treatment for four hours at a temperature between about 2375°-2405° F. followed
by rapid air cooling to ambient, then a heat treatment at 1975. degree. F. for
four hours. This second heat treatment will be usually performed in conjunction
with a coating cycle such as pack aluminide coating or overlay coating. The
final (optional) step is a heat treatment at about 1300° F. for about
twenty-four hours.
Within the broad
ranges presented in Table I a particular relationship should be obeyed to
obtain optimum properties. This relationship is set forth below: ##EQU1## All
values in this equation are weight percent values. The value P given by the
equation is a parameter which predicts the overall merit of the composition.
Compositions with high P values will have high strength in combination with
stability, heat treatability and resistance to oxidation and corrosion.
While it is apparent
that the composition ranges in Table I, particularly the broad composition
range, may encompass specific compositions known in the art, so far as is known
to the inventors, there are no prior art compositions whose P values exceed
approximately 3360. Accordingly, a broad definition of the present invention is
a composition, useful for use in single crystal form, lying within either the
broad, intermediate or preferred ranges of Table I having a P value (as defined
above) in excess of 3360. The invention also concerns single crystal articles
within the composition range having a P value in excess of about 3360. For an
optimum alloy, the most useful composition currently known to the inventors,
the value of P will be slightly in excess of 3940. ##TBL1## (See PDF)
Other prior art
patents relating to single crystals include U.S. Pat. No. 4,402,772 for which
the maximum P value is 2998; U.S. Pat. No. 4,222, 794 for which the maximum P
value is 3329 and U.S. Pat. No. 4,371, 404 for which the maximum P value is
3003.
Table II contains
broad, intermediate and preferred P values for the broad, intermediate and
preferred composition range of Table I. ##TBL2## (See PDF)
Table III sets forth a
series of example compositions which were evaluated in single crystal form. In
Table III alloys B1, B10 and B13 lie outside of the broad range of the present
invention by virtue of their low tantalum levels. Alloy B35 lies outside the
preferred range because it lacks molybdenum.
Table IV sets forth
some of the more significant properties of the single crystal compositions
described in Table III. Also shown in Table III are the P values derived from
the previously described equation for the example compositions set forth in
Table III. Alloy B49 is seen to have the highest P value and an exceptional
combination of properties and is a particularly preferred composition. Data in
Table IV also illustrates the detrimental effect on oxidation behavior which
results from additions of titanium to alloys similar to those of the present
invention. Alloy B10 and alloy B13 are similar except that alloy B10 contains
1% titanium; likewise alloys B1 and B18 are similar except that alloy B1
contains 1% titanium. In coated oxidation resistance, the titanium containing
compositions, B1 and B10, can be seen to have oxidation temperature capabilities
which are 70° F. and 90° F. less than the titanium free B18 and B13
compositions. In uncoated oxidation behavior the B1 and B10 compositions show a
40° F. and 20° F. disadvantage relative to the B18 and B13 compositions
##TBL3## ##TBL4## (See PDF)
The properties of
Alloy B49 will be contrasted with the properties of the alloy described in U.S.
Pat. No. 4,209,348. This prior art composition (nominally 10 Cr, 5 Co, 4 W, 1.5
Ti, 12 Ta, 5 Al, Bal. Ni) is regarded as being among the best of the (prior
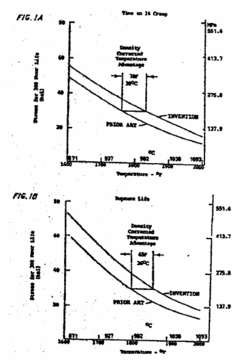
art) single crystal alloys in terms of overall properties. FIGS. 1A and 1B
show, respectively, the stress required to produce 1% creep and rupture in 300
hours as a function of temperature. In FIG. 1A it can be seen that the
invention alloy has a 70° F. temperature advantage on a density corrected basis
over the prior art composition, whereas in FIG. 1B it can be seen that in
rupture life the invention composition has a temperature advantage of about 65°
F. on a density corrected basis over the prior art composition. This means that
in an application where rupture life or creep was the controlling factor, the
invention alloy could be used at a temperature about 65° F., or 70° F. greater
than the prior art alloy while obtaining equivalent life. This improved
temperature capability can be exploited in various ways. Operation at an
increased temperature can produce increased thrust or efficiency. Operating at
a lesser temperature will increase operating life.
FIG. 2 shows a plot of
the stress required to produce 1% creep as a function of the Larson-Miller
parameter which combines time and temperature. It can be seen that the
invention composition has an advantage over the prior art composition, an
advantage which increases with increasing exposure times and/or higher
temperatures. The same comments can be made with respect to FIG. 3 which shows
the stress required to produce rupture as a function of the
previously-mentioned Larson-Miller parameter.
FIG. 4 is a graph
illustrating the oxidation behavior of the prior art composition and the
present invention. The chart shows the hours required to produce a 3 mil deep
oxidation attack on samples tested at different temperatures under cyclic
conditions. The data on the chart was derived from a burner rig test in which
samples were exposed to a high temperature flame, produced by the combustion of
jet fuel, alternating with air cooling. This produces a severe environment
which is representative of turbine engine operation. From FIG. 4 it can be seen
that the present composition has a 65°-70° F. advantage over the prior art in
terms of temperature at which an identical metal loss would be observed.
Alternately, for a given temperature, i.e. 2100. degree. F., the prior art
composition would lose 3 mils of metal in about 43 hours whereas the invention composition
would lose that amount of material in 70 hours, a 63% improvement in oxidation
life.
FIG. 5 is similar to
FIG. 4 except that the samples in FIG. 5 had received a 5 mil coating of an
MCrAlY material whose nominal composition was 22 Co, 17 Cr, 12.5 Al, 0.25 Hf,
0.4 Si, 0.6 Y, balance Ni, prior to testing. The time shown in FIG. 5 is the
time required for coating penetration. It can be seen that the present
invention composition has about a 60° F. temperature advantage over the prior
art. In terms of time to produce coating penetration at 2100. degree. F. the
invention composition requires about a 64% longer exposure time to produce
coating defeat. At 2100° F., the prior art composition was penetrated in about
1400 hours, whereas the present invention composition required about 2300 hours
for coating penetration. The benefits of the MCrAlY coating are apparent when
one compares the 2300 hour figure with the life derived from FIG. 4 at 2100° F.
for an uncoated sample of the present invention of about 70 hours for 3 mils of
attack or less than 200 hours for 5 mils of attack.
The final figure, FIG.
6, illustrates the superiority of the present invention over the prior art
composition in thermal fatigue. Thermal fatigue occurs as a result of stresses
arising from cyclic nonuniform heating. FIG. 6 is a plot showing the total
strain range undergone by the sample in each cycle as a function of the cycles
required to produce failure. The present invention requires about 70% more
cycles to produce failure when compared with the prior art composition. This
corresponds to a 65° F. temperature advantage over the prior art composition.
The final important
material property is corrosion resistance. In this respect the present
invention material has substantially identical performance to that of the prior
art composition. Some relative corrosion data is found in Table IV.
Accordingly, the
invention composition and particularly the preferred embodiment discussed
above, has an advantage of 60°-70. degree. F. over the prior art composition,
the prior art composition which was acknowledged to have the best overall
combination of properties, in all respects except hot corrosion resistance in
which measurement the invention alloy is the equivalent of the prior art
material. Alloy B49 represents a particularly preferred composition. Preferably
no intentional additions of C, B and Zr are made and most preferably these
elements are present in a maximum of 200 ppm C, 30 ppm B, and 75 ppm Zr.
It should be
understood that the invention is not limited to the particular embodiments
shown and described herein, but that various changes and modifications may be
made without departing from the spirit and scope of this novel concept as
defined by the following claims.
TECHNICAL FIELD
This invention relates
to the field of nickel base superalloy compositions adapted to be used in cast
single crystal form and to single crystal articles.
BACKGROUND ART
Nickel base
superalloys are widely used in applications where high stresses must be endured
at elevated temperatures. One such application is the field of gas turbine
engines where nickel base superalloys are widely used especially for blades and
vanes. Demands for improved efficiency and performance have resulted in the
operation of turbine engines at increasingly elevated temperatures placing
extreme demands on the superalloy articles used therein.
One approach which has
been successfully employed to improve the temperature capabilities of nickel
base alloys is to fabricate them in the form of single crystals. Conventionally
prepared metallic materials are comprised of a plurality of grains which are
separated by grain boundaries. The grain boundaries are weak at elevated
temperatures, much weaker than the material within the grains. Through specific
casting techniques nickel base superalloys can be produced in single crystal
form, that is, in the form of articles which have no internal grain boundaries.
U.S. Pat. No. 3,260,505 describes nickel base single crystal superalloy
articles. U.S. Pat. No. 4,116,723 describes a heat treatment method applicable
to a class of nickel single crystal articles. U.S. Pat. No. 4, 209,348
describes a more specific class of single crystal articles and describes a
method for heat treating such articles to improve their elevated temperature
mechanical properties. The nominal composition disclosed in this patent is 10%
chromium, 5% aluminum, 1.5% titanium, 4% tungsten, 12% tantalum, 5% cobalt,
balance essentially nickel. Single crystals of this composition are used
commercially in gas turbine engine applications. This composition is generally
acknowledged to have the best overall combination of properties attainable in a
cast nickel base superalloy article. Another single crystal superalloy
composition is that which is described in U.S. Pat. No. 4,402,772 having a
nominal composition of 6% tantalum, 9% chromium, 5% cobalt, 1% titanium, 1%
molybdenum, 7% tungsten, 5.5% aluminum, 0.15% hafnium, balance essentially
nickel. This material also has desirable properties which are essentially
equivalent to those of the properties in the U.S. Pat. No. 4,209,348 material.
Yet another composition is that described in U.S. Pat. No. 4,222,794 having a
nominal composition of 5.2% chromium, 5.4% aluminum, 1. 1% titanium, 2%
molybdenum, 4.9% tungsten, 6.4% tantalum, 3% rhenium, 0.4% vanadium, balance
essentially nickel.
DISCLOSURE OF
INVENTION
The present invention
comprises a nickel base superalloy composition which can be fabricated into
single crystal articles having an exceptional combination of properties. The
broad composition range is 3-12% chromium, up to 3% molybdenum, 3-10% tungsten,
0-5% rhenium, 6-12% tantalum, 4-7% aluminum, 0-15% cobalt, up to 0.045% carbon,
up to 0.02% boron, up to 0.1% zirconium, up to 0.8% hafnium, up to 2% niobium,
up to 1% vanadium, up to 0.7% titanium, up to 10% of one or more elements selected
from the group consisting of platinum, palladium, ruthenium, rhodium, iridium
and osmium, up to 0.1% yttrium, lanthanum, scandium, cerium, lanthanide or
actinide series of elements, balance essentially nickel. An exemplary and
preferred specific composition within this broad composition range is 5%
chromium, 2% molybdenum, 6% tungsten, 3% rhenium, 9% tantalum, 5.6% aluminum,
10% cobalt, 0.1% hafnium, balance essentially nickel. A compositional
relationship is described which can guide one skilled in the art to the
selection of an optimum composition within the broad range.
Other features and
advantages will be apparent from the specification and claims and from the
accompanying drawings which illustrate an embodiment of the invention.
BRIEF DESCRIPTION OF
DRAWINGS
FIG. 1A is a graph
which illustrates the stress as a function of temperature for the invention
material and a prior art material;
FIG. 1B is a graph
which shows the stress required to produce rupture in 300 hours as a function
of temperature for the invention material and a prior art material;
FIG. 2 is a
Larson-Miller plot of the time to 1% creep compared to a prior art material;
FIG. 3 is a
Larson-Miller plot of the time to rupture compared to a prior art material;
FIG. 4 is a chart
comparing the oxidation behavior of the present invention with that of an
exemplary prior art material;
FIG. 5 is a plot
showing the coated oxidation performance of the present invention compared with
that of a prior art material; and
FIG. 6 is a plot
illustrating the thermo-mechanical fatigue behavior of the invention material
as contrasted with that of the prior art material.
Claims: We claim:
1. A high strength,
heat treatable, stable nickel base superalloy composition particularly suited
for fabrication of single crystal articles, consisting essentially of
* 3-12% Cr, 0-3% Mo, 3-10% W, 0-5% Re, 6-12%
Ta, 4-7% Al, 0-15% Co, 0-0.02% B, 0-0.1% Zr, 0-0.045% C, 0-0.8% Hf, 0-2% Nb,
0-1% V, 0-0.7% Ti, 0-10% (Ru+Rh+Pd+Os+Ir+Pt), 0-0.1% Y, La, Sc, Ce, lanthanide
or actinide series, balance Ni;
* said composition characterized in that where
* -200 Cr+80 Mo-20 Mo**2 -250 Ti**2
-50(TixTa)+15 Cb+200 W-14 W 2 +30 Ta-1.5 Ta**2 +2.5 Co+1200 Al-100 Al**2 +100
Re+1000 Hf-2000 Hf**2 +700 Hf 3 -2000 V-500 C-15000 B-500 Zr=P (where all
elemental values are in weight percent), P is from about 3360 to about 4850.
2. A composition as in
claim 1 in which the value of P is from about 3475 to about 4750.
3. A composition as in
claim 1 in which the value of P is from about 3650 to about 4600.
4. A composition as in
claim 1 having a composition consisting essentially of
* 3-8% Cr, 0.3-3.0% Mo, 3-8% W, 0-5.0% Re,
6-12% Ta, 4.5-6.5% Al, 6-12% Co, 0-0.005% B, 0-0.007% Zr, 0-0.02% C, 0-0.5% Hf,
0-0.4% Ti, 0-10% (Ru+Ir+Pt+Pd+Rh+Os), 0-0.1% Y, La, Ce, Sc, lanthanide or
actinide series, balance Ni, and a value of P (as defined in claim 1) from
about 3450 to about 4750.
5. A composition as in
claim 1 having a composition consisting essentially of
* 4.0-7.5% Cr, 0.5-2.5% Mo, 3.5-7.5% W, 2.5-4%
Re, 8-10% Ta, 5-6% Al, 8-12% Co, 0-0.3% Hf, 0-10% (Ru+Ir+Pt+Pd+Rh+Os),
0.005-0.05% Y, balance Ni, and a value of P as defined in claim 1 from about
3550 to about 4700.
6. A composition as in
claim 5 having a value of P from about 3800 to about 4400.
7. A nickel base
superalloy composition, suited for fabrication into a high strength single
crystal article, consisting essentially of
* 4.0-7.5% Cr, 8-12% Co, 0.05-2.5% Mo, 3.5-7.5%
W, 2.5-4.0% Re, 0-0.04% Ti, 5-6% Al, 8-10% Ta, 0-0.3% Hf, 0-10% of an element
selected from the group consisting of Pt, Pd, Os, Rh, Ru, Ir and mixtures
thereof, 0-0.005% B, 0-0.007% Zr, 0-0.02% C, 0-0.01% Y, La, Ce, Sc, lanthanide
or actinide series, balance essentially Ni;
*
said composition characterized in that where
* -200 Cr+80 Mo-20 Mo 2 -250 Ti 2 -50(TixTa)+15
Cb+200 W-14 W 2 +30 Ta-1.5 Ta 2 +2.5 Co+1200 Al-100 Al 2 +100 Re+1000 Hf-2000
Hf 2 +700 Hf 3 -2000 V-500 C-15000 B-500 Zr=P (where all elemental values are
in weight percent), P is from about 3650 to about 4550.
8. A composition as in
claim 7 which is free from intentional additions of C, B and Zr.
9. A nickel base
superalloy single crystal article consisting essentially of
* 3-12% Cr, 0-3% Mo, 3-10% W, 0-5% Re, 6-12%
Ta, 4-7% Al, 0-15% Co, 0-0.02% B, 0-0.1% Zr, 0-0.045% C, 0-0.8% Hf, 0-2% Nb,
0-1% V, 0-0.7% Ti, 0-10% (Ru+Rh+Pd+Os+Ir+Pt), 0-0.1% Y, La, Sc, Ce, lanthanide
or actinide series, balance Ni;
* said composition characterized in that where
* P=-200 Cr+80 Mo-20 Mo 2 -250 Ti 2 -50
(TixTa)+15 Cb+200 W-14 W 2 +30 Ta-1.5 Ta 2 +2.5 Co+1200 Al-100 Al 2 +100
Re+1000 Hf-2000 Hf 2 +700 Hf 3 -2000 V-500 C-15000 B-500 Zr, P is from about
3360 to about 4850.
10. A single crystal
article as in claim 9 having a composition consisting essentially of
* 3-8% Cr, 0.3-3.0% Mo, 3-8% W, 0-5.0% Re,
6-12% Ta, 4.5-6.5% Al, 6-12% Co, 0-0.005% B, 0-0.007% Zr, 0-0.02% C, 0-0.5% Hf,
0-0.4% Ti, 0-10% (Ru+Ir+Pt+Pd+Rh+Os), 0-0.1% Y, La, Sc, Ce, lanthanide or
actinide series, balance Ni, and a value of P from about 3450 to about 4750.
11. A single crystal
article as in claim 10 having a composition consisting essentially of
* 4.0-7.5% Cr, 0.5-2.5% Mo, 3.5-7.5% W, 2.5-4%
Re, 8-10% Ta, 5-6% Al, 8-12% Co, 0-0.3% Hf, 0-10% (Ru+Ir+Pt+Pd+Rh+Os),
0.005-0.05 Y, balance Ni, and a value of P from about 3550 to 4700.
12. An article as in
claim 9 in which the value of P lies between about 3650 and about 4600.
13. An article as in
claim 10 in which the value of P lies between about 3550 and about 4650.
14. An article as in
claim 10 in which the value of P lies between about 3700 and about 4500.
15. An article as in
claim 11 in which the value of P lies between about 3650 and about 4550.
16. An article as in
claim 11 in which the value of P lies between about 3800 and about 4400.